AstraZeneca enters the Cell therapy space with $200 M takeover of Neogene 👏
Despite the very strong presence in oncology in the past decade, until now, AstraZeneca had been relatively quiet on the cell & gene therapy front. Now, in the span of two weeks, they have clearly signaled their intention to enter the space.
A week ago, info was released on their first in-house CAR-T program [AZD0754] targeting Steap2, an antigen overexpressed in prostate cancer. Now, [2 weeks later] AstraZeneca has made a decisive move, through the acquisition of Neogene, a TCR-T player exploring neoantigen targets in solid tumors, among which KRAS and TP53. The deal is worth up to $320 M, with an upfront payment of $200 M.
The wording in the press release seem to suggest that AstraZeneca has been investing in the space for now 3 years… i guess we can expect to see more from them in the near future (!) “Neogene’s leading (T-cell receptor) discovery capabilities and extensive manufacturing experience complement the cell therapy capability we have built over the last three years”
Source: In house CART program source - Evaluate ; Neogene acquisition source - Fiercebiotech
Bluebird bio sells priority review voucher for $102 M 🧐
With the recent approvals of ZYNTEGLO in beta-thalassemia and SKYSONA in CALD, Bluebird was granted two priority review vouchers, one of which they have now monetized through its sale to Argenx for $102 M.
I only recently became aware of the economics surrounding priority review vouchers… and in my opinion, it’s quite an interesting topic, so here my brief on it:
Priority review vouchers were introduced in 2007 by the US Congress as an incentive mechanism to incentivize R&D efforts for neglected diseases. A “voucher” is obtained upon FDA approval for a new treatment for neglected diseases. This “voucher” then entitles the holder to prioritize the FDA review for any future drug.
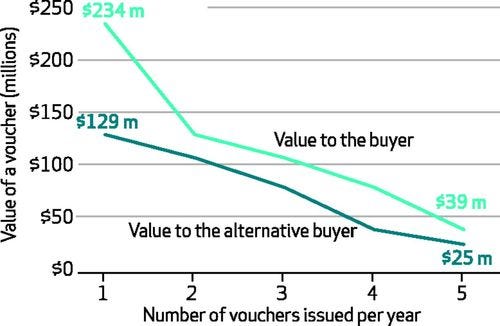
Now where it gets interesting: companies can sell (i.e., monetize) these vouchers. Historically, prices of vouchers range from ~$60 M to ~$350 M. There’s actual studies that look into the value of vouchers, with the main conclusion being that the number of vouchers issued per year is the key driver to the value of the price (see picture below). With these types of numbers, it gets you thinking about whether a truly platform-driven biotech could make a business out of selling priority review vouchers, if they could bring down out-of-pocket R&D costs to <$100 M per asset… food for thought
Source: Health-affairs; Bloomberg
Sana announces 15% reduction in workforce and portfolio reprioritization 😔
Uh oh… When Sana came onto the market last year, they made a splash, breaking the record for highest IPO proceeds: something that was surprising to me considering the pre-clinical stage of the company… The science is certainly very cool, but one has to think about the inherent risks with any pre-IND assets…
One year later, the stock is down ~75% 🤦♂️, the company is letting go of 15% of its workforce 🤦♂️, discontinuing development of a heart failure drug 🤦♂️, and has pushed back its in vivo cell engineered therapy IND submissions into 2023 🤦♂️ (vs 2022… yup, I haven’t forgotten the promise for a 2022 IND for SG295 [link]).
While Sana is grappling on to the IND submission of SC291 this year, this asset is quite frankly not likely to make a splash: its yet another CD19 ex-vivo CAR-T, probably one of ~250 CD19 CAR-Ts in development. No joke… there are 263 CD19 CAR-Ts in development, link
Source: CRI; biopharmadive
Lysogene's lead gene therapy fails pivotal trial in MPS 😔
The company’s top pipeline candidate, LYS-SAF302, failed its phase 2/3 trial among patients with mucopolysaccharidosis type IIIA: while there was statistically significant improvement in cognitive function among the six youngest participants in the trial— kids younger than 30 months, the treatment effect wasn’t seen in the remaining 12 participants "the main cohort". The results come almost a year after Sarepta decided to walk away from a deal to hold ex-Europe rights to LYS-SAF302, a collaboration that cost $15 M in upfront cash in addition to $125 M in biobucks. At the time, Lysogene said the termination had to do in part with “unsuccessful discussions” regarding the transferring of commercial supply of the therapy back to Lysogene.
Source: fierce biotech
*Any views and opinions expressed herein are those of the author (Marco Sabatini) and do not necessarily reflect those of his employer