Cell & Gene Weekly
Reg path forward for Rocket Pharma, Strimvelis in the news and more...
Short week for me as I am on my company’s retreat in Evian. Hence, jumping straight to it. have a good weekend all!
Rocket reaches alignment with FDA on the regulatory path forward for Danon disease asset RP-501
It looks like the FDA may be willing to accept registration of RP-501 for Danon disease based only on an efficacy and safety Ph II trial in 12 patients that includes a pediatric safety run-in (n=2) and leverages natural history data for the comparator arm. If the product were indeed approved solely on this basis (plus the Ph I trial with n = 7 patients [link]), correct me if I am wrong, but I believe this could represent the smallest ever number of patients for an FDA approval… The closest I can think of is probably Zolgensma, which was approved back in 2019, on the basis of a Ph I with 15 patients (START) and a Ph III with 21 patients (STR1VE)… almost double the 7 + 12 patients in this case for Rocket…
What’s more is that it seems the FDA is willing to grant an accelerated approval based on a surrogate endpoint for efficacy (biomarker-based co-primary endpoint consisting of improvements in LAMP2 protein expression and reductions in left ventricular mass)
Talk about uncertainty : this will certainly be an interesting one to follow during the pricing and access negotiations with payers.
Source: BusinessWire
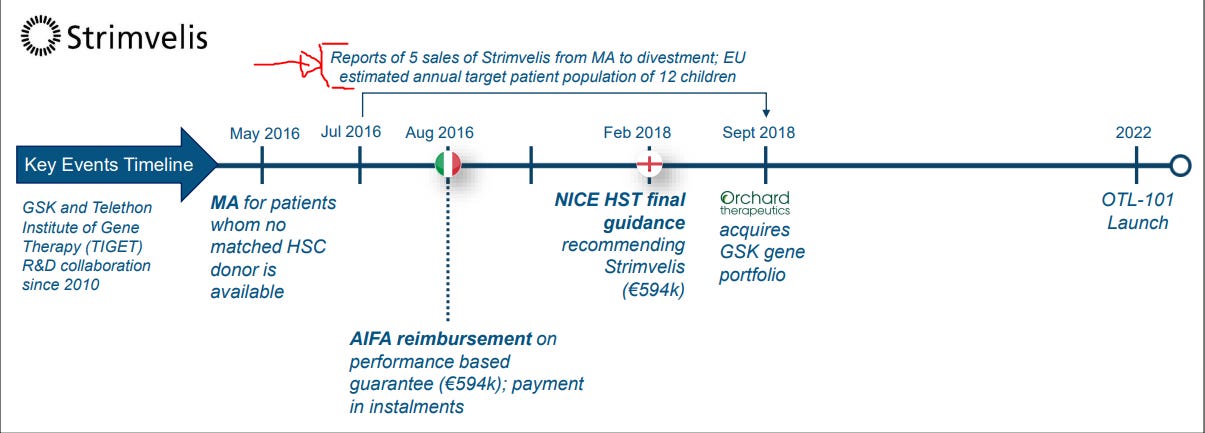
Orchard Tx transfers marketing authorization of Strimvelis to fondazione Telethon
After being initially marketed by GSK in 2016 and transferred to Orchard in 2018, we have now come full circle, “back to the origins”: I’m saying this as the product was originally developed based on research carried out by San Raffaele-Telethon Institute for Gene Therapy, ~15 yrs ago
Strimvelis is one of those classic case studies on the challenges associated with gene therapy commercialization with many “lessons” to be learnt from it. Whilst the product was/is undoubtedly transformational, there are a number of issues related to its usage. One that is quite unique, is the fact that the product has a 6‑hour shelf life and is currently only available in one hospital in the world - Hospital San Raffaele Telethon Institute for Gene Therapy in Milan. This means that to get access to it, people need to travel to this hospital for treatment. Despite of this, it has received approval in a few countries; as an example for the UK, NICE approved the product stating that it can provide “NHS funding for travel and accommodation costs for people having Strimvelis and their families”.
To add to that, the disease is so rare, that it is probably not even commercially viable to justify and set up a second center for administration. It is estimated that in the EU, the target population is 12 patients (only!!!), making commercialization certainly challenging… Working backwards from the press release that states that 45 patients have been treated to date [incl the 24 in clinical trials], this would imply that in its 7 years on the market, it has “only” been administered in 21 patients commercially? Ouch.
Source: P4A presentation; NICE website; PRNewswire
Cellares and Lyell to Evaluate Automated Manufacturing of Lyell's CAR T-Cell Therapy on Cellares' Cell Shuttle Platform
Second time in 3 weeks that i’m covering Cellares, so please skip the overview below if you already saw this in a previous edition. Last time it was a collab with BMS, this time, it’s with Lyell
Cellares and Lyell have agreed on a proof-of-concept technology transfer process for the manufacture of Lyell's LYL797 (a ROR1 CAR T for treatment of solid tumors) using the Cell Shuttle.
Cellares’ “Cell Shuttle” technology is actually quite cool (if indeed able to deliver what they boast about…). They seem to have a truly closed and automated end-to-end equipment that would allow for scalability, i.e., enabling 1000’s of individual patient doses per year.
If you want to hear from the Cellares CEO directly on building out the “Cell Shuttle” to automate cell therapy manufacturing, here is a good interview from BiotechTV: https://www.biotechtv.com/post/cellares-august-30-2023
Source: PRNewswire
Good find
William Blair have published a new piece on In Vivo CAR-T Landscape, considering the various modalities (LV, LNP, nLNP, etc). I tried to embed the X (Twitter) post here, but was unable to, so sharing here the source directly from which I found this - source
Quick take news
Novartis Drops Eye Disease Gene Therapy from $800M Gyroscope Acquisition | BioSpace [Source]
2seventy bio Announces Strategic Restructuring to Prioritize Growth of Abecma, Streamline Pipeline Advancement and Preserve Financial Runway [Source]
*Any views and opinions expressed herein are those of the author (Marco Sabatini) and do not necessarily reflect those of his employer
Have I missed anything? Is there something you would like to hear more about? let me know !