Some of the usual themes covered in this edition: CAR-Ts in autoimmune, pipeline reprioritizations, and some regulatory updates, including formal approval of Casgevy in Europe (from EC), following positive CHMP in late 2023.
Enjoy the read and have a good weekend ahead
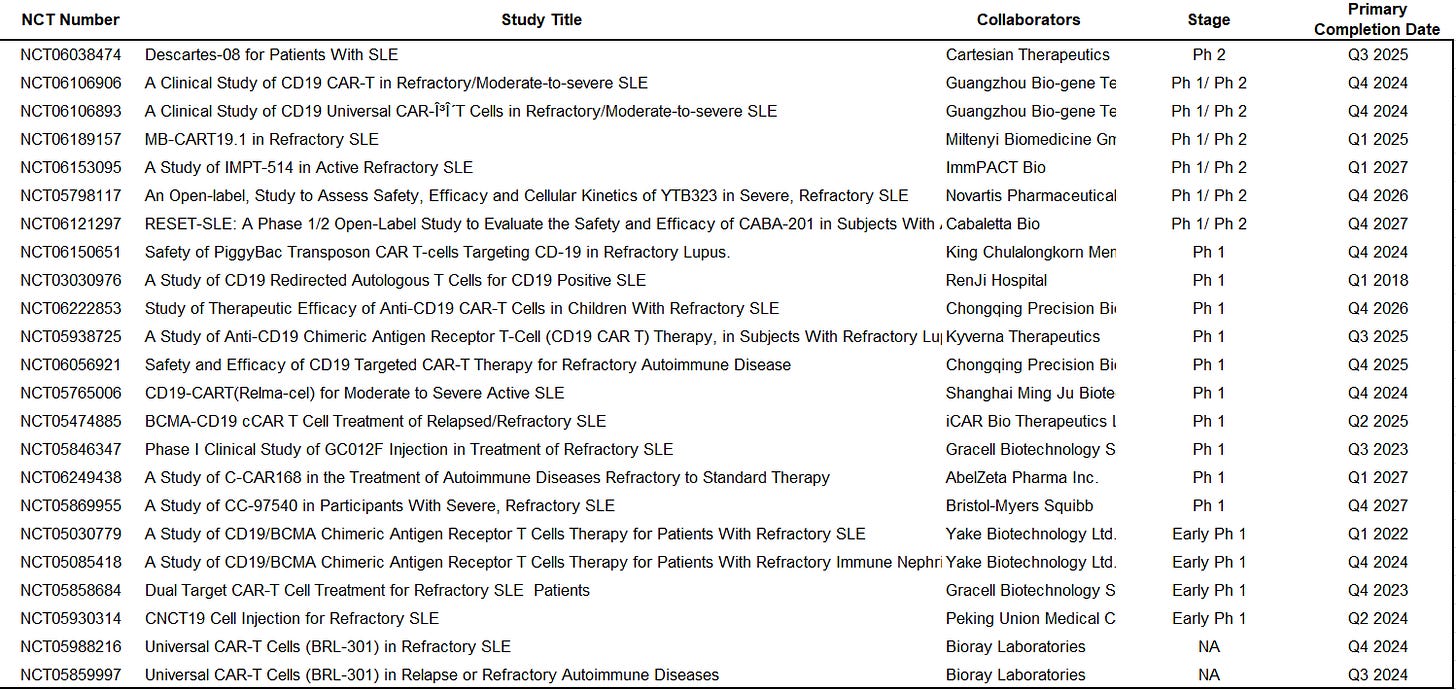
All aboard the CD19 CAR-T lupus bandwagon ! Atara adds itself to the long list of cell therapy players pursuing Lupus
Atara announced submission of an IND application to the FDA for the use of ATA3219, an allo CD19 CAR-T, for the treatment of SLE with kidney involvement (lupus nephritis). As I’ve noted a few times now, the list of CAR-Ts moving into autoimmune is getting very long... The usual phenomenon of therapeutic herding.
For the fun of it, I did a quick scan on clinicaltrials.gov to check out how many players are pursuing CAR-Ts in SLE… 23 registered CAR-T clinical trials in SLE to date, mostly in Ph 1 or Ph 1/2. Yikes.
P.S. dont forget that on top of the clinical stage assets above, there’s a number of companies that have signaled intent to pursue / submit IND’s to go into the clinic, e.g., NKarta [link], Fate therapeutics [link]
Source: Atara, clinicaltrials.gov
After their announced reprioritization last year, Taysha transfers rights to 3 programs back to originating institutions
Taysha is one of those interesting biotech case studies: in 2020 it was labelled “fastest seed to IPO” company in biotech history. At IPO, Taysha boasted 18 programs in their pipeline (albeit most of them being pre-clinical). Fast forward to 2022-23: reduction(s) in workforce, pipeline reprioritization(s) and hopeful patients / patient families stripped of hope.
In the latest announcement, Taysha is (partially) redeeming itself (to families and originators of the science), transferring rights for a number of their deprioritized programs to the originating institutions:
Rights to TSHA-101, TSHA-104 and TSHA-112 have been transferred to their originating institutions
Process has been started to transfer the IND application and associated clinical trial materials for TSHA-120 in giant axonal neuropathy to the National Institute for Neurological Disorders and Stroke
Provided investigational clinical trial material for TSHA-118 in CLN1 to support an individual-patient investigator-initiated IND request from RUSH University Medical Center for the treatment of a patient with CLN1 disease
So What? A moral / ethical win - an important move to ensure that science continues to advance, and patient benefit from potentially transformation therapies… i.e., putting patients at the forefront of every decision
Side note - If you want to read more about the alternative approaches to drug discovery for ultra-rare diseases, I would recommend reading Perlara’s Cure Odyssey, which provides a powerful perspective on the “anti-portfolio of pharma” [link]
Source: Taysha website; [image, credit of Ethan Perlstein on Perlara’s Cure Odyssey’s]
Quick take news:
Sana Biotechnology Announces Publication of Preclinical Diabetes Data in Cell Stem Cell Demonstrating Insulin Independence Following Transplantation of Hypoimmune Allogeneic Primary Islet Cells Without Immunosuppression in a Diabetic NHP [source]
Precision BioSciences Receives Pre-IND Feedback from US FDA for PBGENE-HBV as it Advances Towards Clinical Readiness [source]
Genenta Demonstrated Reprogramming of the Tumor Microenvironment in GBM Patients, Paving the Way for Innovative Treatments of Solid Tumors [source]
Intellia Therapeutics and ReCode Therapeutics Announce Strategic Collaboration to Develop Novel Gene Editing Therapies for Cystic Fibrosis [source]
Bayer's Ask Bio unit starts Phase II heart failure gene therapy [source]
Regulatory quick take news:
CRISPR gene therapy Casgevy secures approval in Europe [source]
FDA has extended the priority review period for Rocket Pharma’s gene therapy Kresladi for the treatment of LAD-I by three months to June 30 to give the agency more time to review certain information. The issue seems to be CMC related and probably not a major source of concern given; there’s precedent for this kind of delay in C&G space (e.g., Krystal Bio 3 month delay last year) + FDA has confirmed no AdComm is needed
*Any views and opinions expressed herein are those of the author (Marco Sabatini) and do not necessarily reflect those of his employer