CAR-Ts under scrutiny for early label expansion in MM
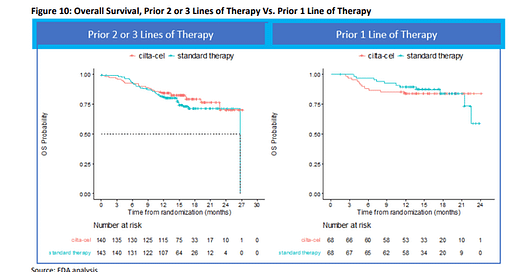
The FDA is holding an adcom for Carvykti and Abecma today looking into the increased rates of death for the CAR-Ts compared with standard drug combinations in the first months of their respective Ph 3 trials. The concern looks to be more pronounced for Abecma vs Carvykti, given that the later flips this OS disadvantage after the 11 month (see image below), especially in the 3L and 4L patient population (left hand image below).
As a justification for the early deaths, BMS cited the fact that some patients didn’t live to receive Abecma (i.e., manufacturing, QC and re-infusion process being too long, especially in controlled clinical trial setting) while J&J, pointed to bridging therapy as a possible culprit. Either way, this puts into question the benefit of these CAR-T’s in this early setting, likely requiring the companies to product more data to convince regulators, payers and physicians of the value in 2L or 3L+. By end of day, we’ll at least have a clearer view on where the FDA might land on the benefit : risk assessment for the two therapies …
Source: FDA Carvykti adcom brief; FDA Abecma adcom brief
BMS scores approval for Breyanzi in CLL / SLL
FDA granted an accelerated approval for 2L+ chronic lymphocytic leukemia (CLL) or small lymphocytic lymphoma (SLL). To be eligible for the CD19-targeted CAR-T med, a patient must have tried a BTK drug (e.g., Brukinsa) and a BCL2 inhibitor (e.g., Venclexta). Breyanzi has a relatively lower ORR (45%) vs small molecule competitor Jaypirca, but 20% CRs and a long median duration of response, reaching 35.3 mo. The unique CLL/SLL label now gives Breyanzi an edge vs Yescarta :)
Source: fiercepharma
GLP-1 Gene therapy? New pre-clinical data for Rejuva
Fractyl health published preclinical findings (mice data) for its Rejuva gene therapy platform showing greater blood sugar reduction and weight loss vs vehicle with chronic semaglutide, i.e., 50% blood sugar lowering and 11% weight loss vs 32% and 2% respectively for semaglutide. The data is very early stage but interesting, especially in the context of the current GLP-1 craze. What does a one-time therapy of this sort overcome?
High discontinuation rates of GLP-1s… and the fact that if you stop you may regain 2/3 of the weight lost… (this seems to be a real issue, with real world studies suggesting 40-50% patients discontinuing within ~1 yr…)
Long term tolerance to GLP-1s? Whilst this is a topic needing further confirmation, one could imagine tolerance to treatment building up…
What’s neat about this is that it’s not necessarily just a GLP-1 replacement, but a hybrid of bariatric surgery and GLP-1 admin; and the hope is that it would be a [much] safer approach vs bariatric surgery, coming with the benefits of long term GLP-1 —> a “lasik for obesity”. Keen to follow this moving into the clinic trial setting !
Source: BMJ RWE on GLP-1 use in UK; PPA RWE on GLP-1 in US; Fractyl IR presentation; Fractyl press release
I couldn’t resist…
Quick Take news
Intrathecal bivalent CAR T cells targeting EGFR and IL13Rα2 in recurrent glioblastoma: phase 1 trial interim results; no ORRs but reductions in enhancement and tumor size at early MRI timepoints observed in all six patients [source]
Cellares Set to Launch Good Manufacturing Practice–Compliant Cell Shuttle Manufacturing Platform [source]
bluebird bio Announces First Outcomes-Based Agreement with Medicaid for Sickle Cell Disease Gene Therapy [source]
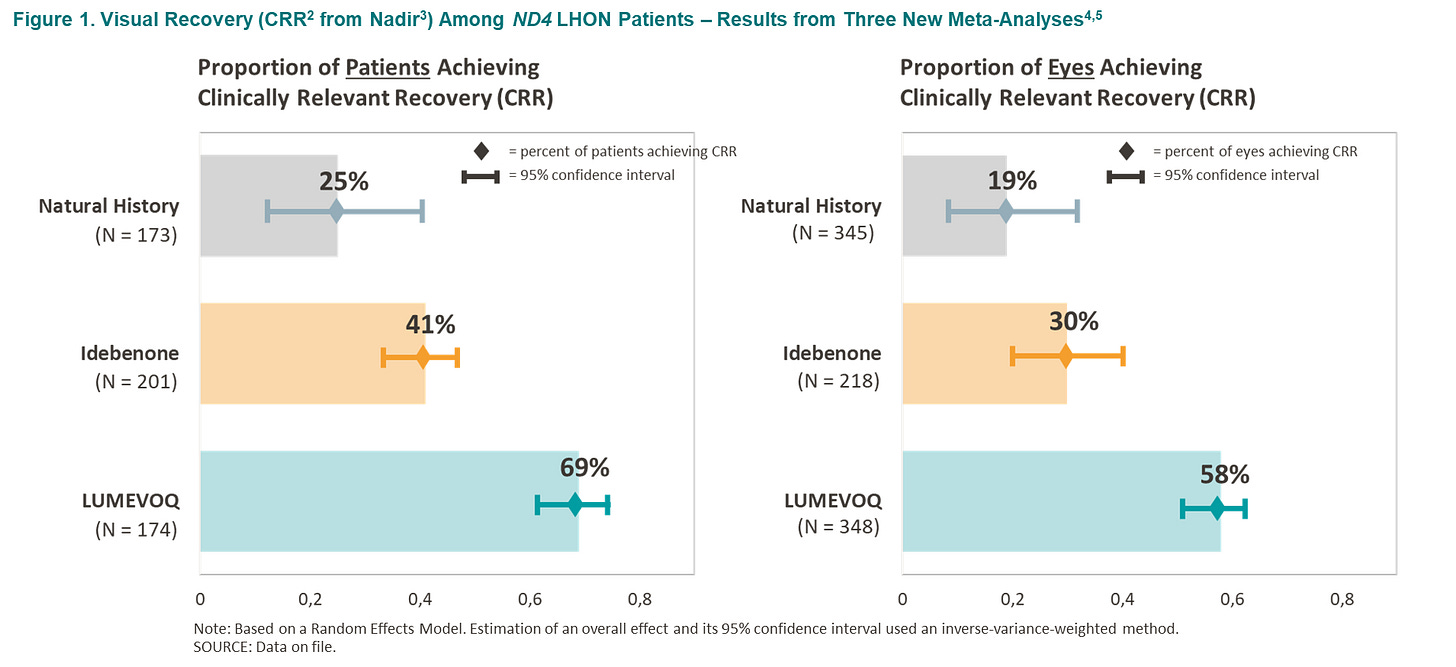
GenSight Biologics Announces Initial Results from New Meta-Analyses on Visual Outcomes with LUMEVOQ® Gene Therapy at NANOS 2024 [source]
*Any views and opinions expressed herein are those of the author (Marco Sabatini) and do not necessarily reflect those of his employer
Have I missed anything? Is there something you would like to hear more about? let me know !




Lots of interesting news in the space.... congrats to Breyanzi team.