Quiet week in the Cell & Gene space, hence not much to add other than … Have a restful weekend all!
CAR-T clinical trials set to start in Multiple Sclerosis
The CAR-T race in the autoimmune space is heating up, and while we’re already starting to see some drop outs in SLE (e.g., Galapagos dropping their CD19 CAR-T program in rSLE last week), were starting to see a broadening in the scope of indications. As of end of Feb 2024, 3 trials had been posted in multiple sclerosis:
Bristol-Myers Squibb Company: CD-19 CAR-T using NEX-T platform → already recruiting !
RenJi Hospital: dual BCMA / CD20 CAR-T
Kyverna: CD19 CAR-T
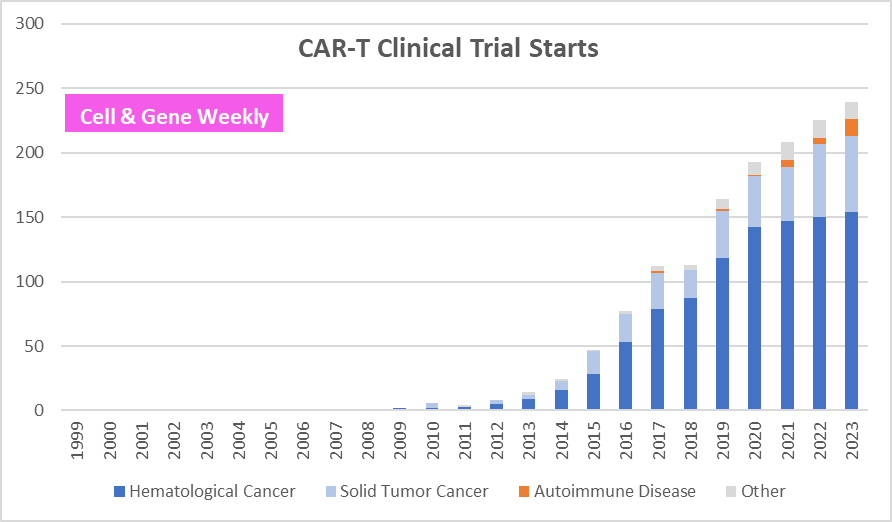
In the spirit of curiosity, I downloaded from clinicaltrials.gov all entries relating to CAR-T over a 20 year period and used AI (Julius.ai*) to sort the data according to therapeutic area. Some notable insights from the exercise:
There’s quite a few trials in autoimmune beyond SLE, e.g., Myasthenia Gravis, Idiopathic Inflammatory Myopathies, Crohn Disease, Ulcerative Colitis, Sjogren's Syndrome, Scleroderma, Autoimmune Hemolytic Anemia
The first CAR-T trials in autoimmune dates back from 2017-19… not so new after all
>50% of the autoimmune CAR-T trials originate from China, with Zhejiang University being the leader (by # of trials registered on clinicaltrials.gov)
Below is a visual of the data that I (i.e., Julius.ai*) analyzed
Source: clinicaltrials.gov, Julius.ai, nature
*While Julius.ai is great in principle, not sure I saved much time, given the number of iterations I had to go through to get a decent output...
Status check on the commercialization of Gene therapies
Sarepta and Krystal bio posted Q4 2023 results: both launches are off to a very good start !! (unlike those of Roctavian, Hemgenix, Skysona, Zynteglo, etc. that have had a relatively slow ramp up… unsurprisingly given the complex ex-vivo manufacturing)
Sarepta: Elevidys QoQ increase from $69.1 m in Q3 ‘23 to $131.3 m in Q4 ‘23 [source] —> 👏👏 A remarkable launch, which reminds me of that of Zolgensma back in 2019. This is what Dallan Murray, the CCO, had to say about it:
“We're extremely pleased with the launch execution, exceeding our own lofty expectations. In fact, the $200 million in net product revenue surpassed the combined 2023 revenue of the other five gene therapy launches from the past 18 months.” [source]
Krystal bio: Vyjuvek QoQ increase from $8.6 m Q3 ‘23 to $42.1 m in Q4 ‘23 [source] —> 👏👏 Another remarkable launch, with Krish Krishnan, CEO of Krystal, noting that “our commercial launch is proceeding exceptionally well” [source]. Looks like this could reach blockbuster status at peak ?
Quick take news:
EU Panel Backs J&J, Legend’s Carvykti for Second-Line Multiple Myeloma [source]
BeiGene ends pact with Shoreline, its ‘entry into cell therapy’ [source]
Friedreich’s ataxia gene therapy candidate selected under Voyager and Neurocrine collaboration [source]
Miltenyi Biotec launches India operations, to open Centre in Hyderabad [source]
Blue Gen Therapeutics Foundation Secures Exclusive License for Promising Gene Therapy Programs from Adverum Biotech [source]
Interesting reads
Fetal surgery expert Tippi MacKenzie shifts to in-utero gene therapy [source]
Phage-assisted evolution of highly active cytosine base editors with enhanced selectivity and minimal sequence context preference [source]
*Any views and opinions expressed herein are those of the author (Marco Sabatini) and do not necessarily reflect those of his employer
Have I missed anything? Is there something you would like to hear more about? let me know !